Science
Physics (Middle School)
Scientific Measurement and Data; Scalars and vectors; Forces and Motion (Newton’s Laws, Gravity, Friction, Contact Forces, Kinematics); Energy (Kinetic & Potential Energy, Energy Transformation & Conservation); Waves (Properties of waves, Sound Waves, Electromagnetic Waves, Optics); Electricity & Magnetism (Static Electricity, Simple Circuits, Magnetic Fields & Forces); Matter and its Physical Properties; Heat and Thermal Energy; Engineering and Simple Machines.

Physics (High School)
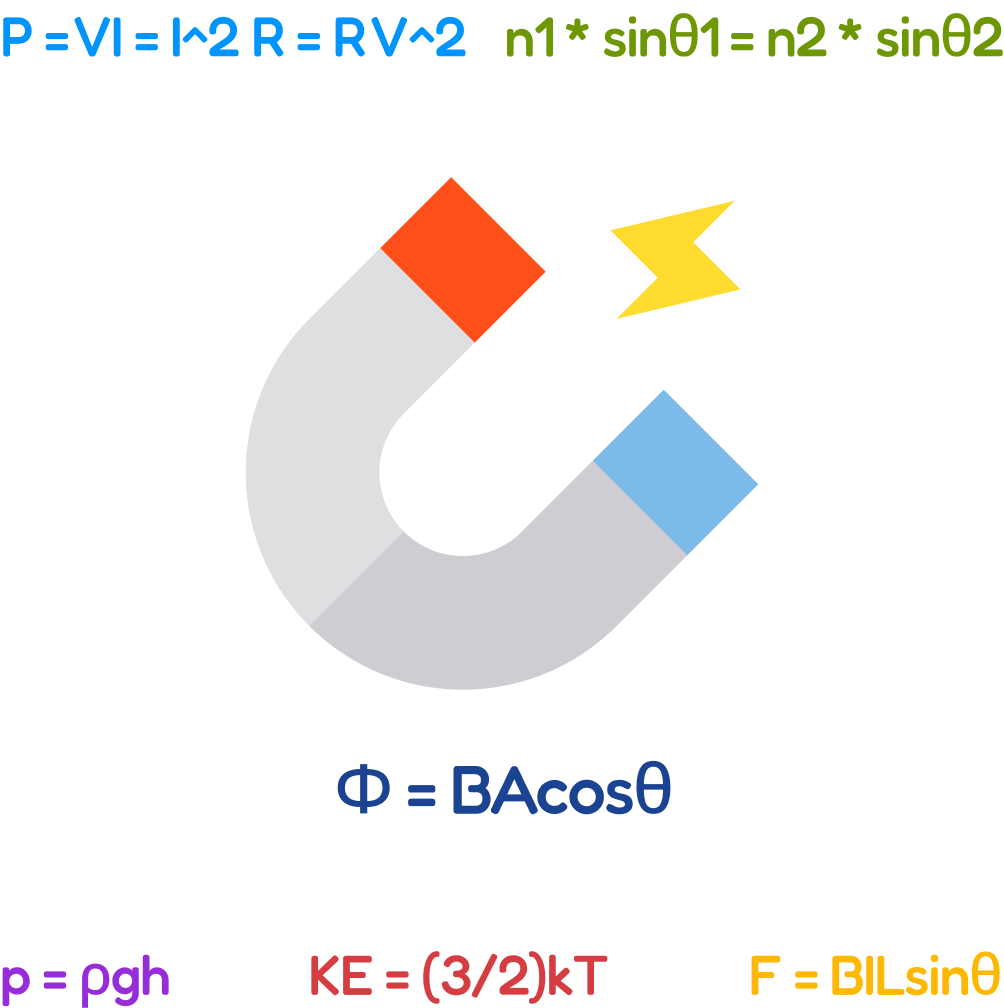
Scientific Measurement and Modeling; Kinematics (Motion); Dynamics (Newton’s Laws, forces, gravity, friction); Work, Energy, and Power; Momentum and Collisions; Rotational Motion (Torque, angular momentum, circular motion); Waves and Optics (Wave properties, Sound waves, Doppler effect, Light, Geometric Optics); Electricity and Circuits (charge, fields, potential, Direct Current (DC), Ohm’s Law); Magnetism and Electromagnetism, Thermodynamics (Heat, temperature, kinetic theory, gas laws); Fluid Mechanics (Density, buoyancy, pressure).
Chemistry (Middle School)
Building blocks of matter (atoms, molecules, elements), Periodic Table, States of matter (solids, liquids, gases), basic changes in matter, hands-on exploration of chemical reactions, density, acids/bases, and energy, atomic structure (protons, neutrons, electrons), Chemical Bonding, Chemical Formulas and Chemical Compounds, Chemical Equations and Reactions.

Chemistry (High School)
Scientific process and Measurement, Properties of matter, physical/chemical changes, states of matter (solid, liquid, gas), atomic structure (protons, neutrons, electrons, isotopes), electron configuration, quantum numbers, Periodic Table, Trends (radius, ionization energy, electronegativity), groups, periods, valence electrons, Lewis structures, Bonding ( Ionic, covalent, metallic bonds, molecular geometry, intermolecular forces), Molar mass, Avogadro’s number, mole calculations, empirical/molecular formulas, Types of chemical reactions, writing and balancing equations, stoichiometry (mole ratios, limiting reactants), States of Matter & Gases, Kinetic Molecular Theory, ideal gas law, gas properties, Thermochemistry, Heat, Temperature, enthalpy, exothermic/endothermic processes, calorimetry, Kinetics and Equilibrium, Reaction rates, Le Châtelier’s Principle, equilibrium constants, Acids & Bases, Definitions (Arrhenius, Brønsted-Lowry), pH scale, Oxidation-Reduction (Redox) reactions, Nuclear Chemistry (radioactivity, decay), Organic Chemistry basics. .

